The Structural Framework of Healthcare and Medical Research
The journey from a nascent scientific idea to a life-saving medical intervention is a complex, multi-stage process, meticulously structured to ensure safety, efficacy, and reproducibility. This structural framework underpins all advancements in healthcare and medical research, guiding discoveries from the laboratory bench to the patient’s bedside.
It begins with basic research, often termed “bench science” or fundamental research. This stage is driven by curiosity and the pursuit of knowledge for its own sake, exploring the fundamental mechanisms of biology, disease pathology, and human physiology. Researchers delve into cellular processes, genetic codes, and molecular interactions, laying the groundwork without an immediate therapeutic goal in mind. The insights gained here are crucial, as they often uncover the targets for future treatments.
Following basic research, successful findings transition into pre-clinical studies. Here, the focus shifts to evaluating potential therapies in laboratory settings, typically using cell cultures and animal models. This stage aims to assess the safety, dosage, and initial efficacy of new drugs, devices, or procedures before they are introduced to human subjects. Rigorous testing at this phase helps identify potential toxicities and provides critical data to justify moving to human trials.
The most visible and regulated part of this journey is clinical research, which involves studying interventions in human volunteers. This phase is divided into several stages, each with specific objectives:
- Phase 0: Exploratory, small-scale studies (10-15 volunteers) with sub-therapeutic doses to understand drug behavior (pharmacokinetics and pharmacodynamics) in humans.
- Phase I: Focuses on safety and dosage in a small group of healthy volunteers (20-100) or patients with the condition.
- Phase II: Evaluates efficacy and further assesses safety in a larger group of patients (100-300) to determine optimal dosing and side effects.
- Phase III: Compares the new treatment to existing standard treatments or a placebo in a large, diverse patient population (300-3,000 or more) to confirm efficacy, monitor side effects, and gather information for safe use.
- Phase IV: Post-marketing surveillance after a treatment is approved and on the market. This phase monitors long-term effects, optimal use, and rare side effects in the general population.
Major institutions globally play indispensable roles in advancing this research pipeline. The National Institutes of Health (NIH), as the largest biomedical research agency in the world, funds a vast array of studies, from basic science to clinical trials, driving progress across hundreds of health-related areas. Institutions like Harvard Medical School and the Mayo Clinic are powerhouses of both research and clinical care, integrating cutting-edge discoveries directly into patient treatment. Harvard Medical School, for instance, is renowned for its pioneering work in areas like genomics and personalized medicine, while Mayo Clinic is celebrated for its patient-centered research model, translating laboratory findings into clinical practice with remarkable speed.
Prestigious medical journals such as The New England Journal of Medicine (NEJM), Journal of the American Medical Association (JAMA), and Nature Medicine serve as critical platforms for disseminating these advancements. They rigorously peer-review and publish groundbreaking studies, shaping medical practice and informing healthcare policy worldwide. These journals are not just repositories of information; they are arbiters of scientific quality and integrity, ensuring that only the most robust and impactful research reaches the global scientific community.
Funding Dynamics in Healthcare and Medical Research
The scale and complexity of modern medical research demand substantial financial investment, drawing from a diverse array of sources. Global research and development (R&D) spending reached an estimated $2.9 trillion in 2024, reflecting a worldwide commitment to innovation. The United States and China lead this investment, with both countries spending approximately $780 billion each. Asia collectively accounts for roughly 45% of global R&D spending, followed by North America and Europe.
In the biomedical sector specifically, expenditures are immense. In 2012, global biomedical R&D totaled $268.4 billion, with the United States contributing a significant 44.5% ($119.3 billion). This funding primarily comes from three major avenues: government grants, industry investment, and private philanthropy.
Government grants, particularly from agencies like the NIH, are foundational. The NIH’s support for biomedical research grew from $11 billion in 1995 to $27 billion by 2010, demonstrating a sustained commitment to public health. These grants often support basic research and early-stage translational studies, where the commercial viability is still uncertain. Initiatives like the 21st Century Cures Act aim to accelerate the discovery, development, and delivery of new treatments, while the NIH Common Fund supports high-impact, trans-NIH programs that address critical gaps in biomedical research.
Industry investment, primarily from pharmaceutical and biotechnology companies, has become the largest single source of biomedical research funding. Recent data indicates that industry sources funded 62% of biomedical research, a significant increase from 32% in 1980. This shift reflects a growing focus on late-stage clinical trials and product development, where the potential for commercial return is higher. While industry funding is vital for bringing therapies to market, it also introduces ethical considerations regarding conflicts of interest, which we will explore later.
Private philanthropy from foundations, charities, and individual donors also plays a crucial role, often funding niche areas, rare diseases, or innovative approaches that may not attract large government or industry investment. This diverse funding landscape ensures a broad spectrum of research can be pursued, from fundamental discoveries to applied clinical solutions.
Opportunities for Early-Career Researchers
The vibrant field of medical research offers numerous pathways for aspiring scientists and clinicians. Cultivating the next generation of researchers is paramount to sustaining innovation and addressing future health challenges.
For high school students with a burgeoning interest in science, programs like Harvard Medical School’s pre-college courses provide an invaluable introduction. These courses, offering approximately 30 hours of instruction and activities, immerse students in college-level content, covering topics from genomics to therapeutic development. They often include capstone projects and mentorship, allowing students to design studies and engage with real-world medical research scenarios. This early exposure can be pivotal in solidifying career aspirations in STEM.
At the collegiate and postgraduate levels, the opportunities expand significantly. The NIH offers a wide array of training programs and fellowships designed to support students and early-career researchers at various stages. These include undergraduate internships, graduate research fellowships, and postdoctoral training programs that provide hands-on experience in leading laboratories across the country. These programs are instrumental in developing the specialized skills and knowledge required for a successful career in biomedical research.
Beyond formal training, postdoctoral fellowships are critical for researchers transitioning from doctoral studies to independent research careers. These positions allow individuals to gain further specialized expertise, lead their own projects, and publish their work, which is essential for securing faculty positions or leadership roles in industry.
The broader landscape of biomedical career development also includes opportunities in clinical research coordination, bioinformatics, medical writing, and regulatory affairs. These roles, while not always at the bench, are integral to the research ecosystem. Mentorship, networking, and continuous learning are key components of success in this dynamic field, enabling early-career professionals to contribute meaningfully to advancements in healthcare.
Technological Frontiers and Molecular Breakthroughs
The 21st century has ushered in an era of unprecedented technological innovation, profoundly transforming the landscape of medical research. Advanced tools and methodologies are allowing us to probe the complexities of human health and disease with remarkable precision, leading to breakthroughs that were once unimaginable.
Artificial Intelligence (AI) and machine learning (ML) are at the forefront of this revolution, processing vast amounts of data to uncover patterns, predict outcomes, and optimize interventions. Coupled with big data analytics, AI can sift through genomic, clinical, and lifestyle information to identify novel disease biomarkers and therapeutic targets.
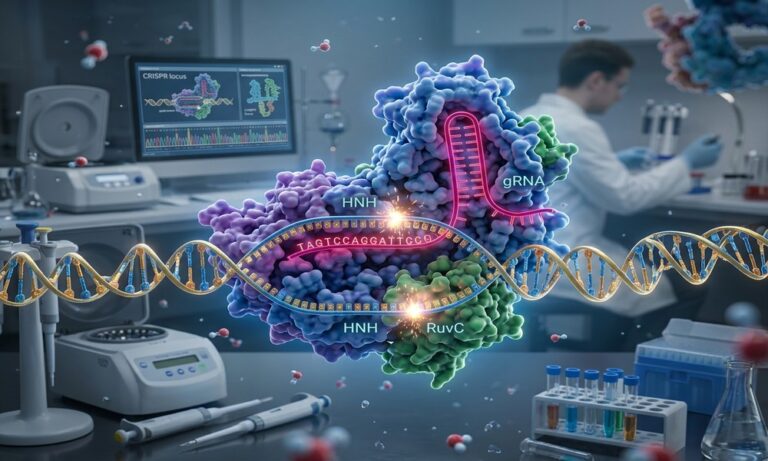
CRISPR gene editing technology, often visualized as a molecular scissor, represents a paradigm shift in our ability to manipulate genetic material. This precise tool allows scientists to cut, add, or alter DNA sequences with unprecedented accuracy, opening doors for correcting genetic defects that cause diseases like cystic fibrosis, sickle cell anemia, and Huntington’s disease. The potential of CRISPR extends beyond single-gene disorders, offering new avenues for cancer immunotherapy and infectious disease resistance.
Genomic sequencing has become faster and more affordable, enabling comprehensive analysis of an individual’s entire genetic makeup. This capability is foundational to personalized medicine, allowing treatments to be tailored to a patient’s unique genetic profile. The integration of these technologies is not just accelerating discovery but also making medicine more predictive, preventive, and precise.
In the realm of therapeutic development, the understanding of complex biological molecules is advancing rapidly. For instance, the development and application of Biogenix medical peptides are showing promise in targeted therapies. These small chains of amino acids can be engineered to interact with specific receptors or pathways in the body, offering highly selective treatments with potentially fewer side effects than traditional drugs. Research into these specialized molecules exemplifies how molecular breakthroughs are driving the creation of novel and more effective medical interventions.
The Role of AI in Healthcare and Medical Research
Artificial intelligence is rapidly becoming an indispensable tool across the entire spectrum of healthcare and medical research, from optimizing administrative tasks to revolutionizing patient care and drug discovery. Its capacity to analyze vast datasets far exceeds human capabilities, leading to insights that were previously unattainable.
In diagnostics, AI algorithms can interpret medical images such as X-rays, MRIs, and CT scans with remarkable accuracy, often identifying subtle anomalies that might be missed by the human eye. This capability is improving the early detection of diseases like cancer and retinal conditions, leading to more timely and effective interventions.
Beyond diagnostics, AI is enhancing the efficiency of clinical practice. AI scribes, for example, are transforming how clinicians document patient visits. These intelligent systems can listen to patient-doctor conversations, transcribe them, and automatically populate electronic health records. Multisite studies have shown that the adoption of AI-powered scribes can significantly alter clinician time expenditure, potentially freeing up doctors to spend more time with patients rather than on administrative tasks, and in some cases, even increasing visit quantity by streamlining workflows. Ambient AI, which passively observes and assists, is contributing to the “quintuple aim” in healthcare by improving patient experience, provider well-being, health outcomes, and health equity, all while reducing costs.
Predictive analytics powered by AI are being used to forecast disease outbreaks, identify patients at high risk for certain conditions, and even predict responses to specific treatments. This allows for proactive interventions and more personalized care plans. In neonatal care, AI models are being developed for precision nutrition, adapting feeding regimens for premature infants based on real-time physiological data, leading to improved growth and developmental outcomes.
The integration of AI into medical research is not just about automation; it’s about augmenting human intelligence, enabling researchers to ask more complex questions and find answers faster, ultimately accelerating the pace of medical discovery.
Personalized Medicine and Genomics
Personalized medicine, often referred to as precision medicine, represents a paradigm shift in healthcare, moving away from a “one-size-fits-all” approach to one that tailors medical decisions, treatments, practices, and products to the individual patient. At its core, this approach is deeply intertwined with genomics and the understanding of genetic diversity.
The All of Us Research Program in the United States is a monumental initiative collecting health data from a million or more volunteers. This program aims to build one of the largest and most diverse health databases globally, encompassing genetic information, lifestyle data, and environmental exposures. By studying such a diverse cohort, researchers can identify how individual variability in genes, environment, and lifestyle influences health and disease, paving the way for truly personalized interventions.
Understanding an individual’s genetic makeup allows for more targeted therapies. For example, in oncology, genetic sequencing can identify specific mutations in a patient’s tumor, guiding oncologists to select drugs that are most likely to be effective against that particular genetic profile. This not only improves treatment success rates but also minimizes exposure to ineffective therapies and their associated side effects.
Beyond genes, personalized medicine also considers the intricate details of biological molecules. Advances in structural biology, such as visualizing protein 3D modeling, allow scientists to understand the precise shape and function of proteins. This knowledge is crucial for designing targeted therapeutics that can specifically interact with disease-causing proteins, offering highly effective treatments. Research into cellular processes, such as mitochondrial metabolism, is also revealing new insights into aging, cancer, and neurodegenerative diseases, opening avenues for interventions that can modulate these fundamental biological pathways.
For conditions like rare disease registries, genomic information is particularly transformative. Many rare diseases have a genetic basis, and accurate diagnosis through sequencing can be life-changing for patients and their families, enabling access to specific treatments or clinical trials. The ability to analyze and interpret vast amounts of genomic data, combined with other health information, is making personalized medicine a reality, promising a future where healthcare is as unique as each individual.
Advancements in Clinical Dentistry and Chronic Disease
Medical research extends its profound impact across a vast spectrum of health, from the intricate details of oral health to the complex challenges of chronic diseases like cancer, heart conditions, and neurological disorders. Significant strides are being made, promising better prevention, diagnosis, and treatment for millions worldwide.
In the realm of oral health, the integration of advanced technologies and research findings is transforming clinical dentistry. Modern dental practices are increasingly leveraging insights from medical research to offer more effective and less invasive treatments. This includes advancements in biomaterials, pain management, and diagnostic imaging that improve patient outcomes. For those seeking comprehensive care, understanding the landscape of Advanced dental healthcare is crucial, as it encompasses everything from preventative measures to complex reconstructive procedures, all informed by ongoing scientific discovery.
Chronic diseases, which account for a major portion of global morbidity and mortality, are a primary focus of medical research. Initiatives like the Cancer Moonshot aim to accelerate cancer research to make more therapies available to more patients, reducing cancer incidence and improving survival rates. Similarly, the BRAIN Initiative is dedicated to revolutionizing our understanding of the human brain, seeking new ways to treat, cure, and prevent devastating neurological disorders.
Research into Alzheimer’s biomarkers is yielding promising results, with studies showing that certain blood biomarkers can predict dementia up to 16 years before onset. This early detection is critical for future interventions that could slow or prevent disease progression.
In cardiovascular health, breakthroughs are improving the management of conditions affecting millions. From novel drugs to advanced surgical techniques, research continues to enhance our ability to prevent heart attacks, strokes, and other cardiovascular events. The synergy between medical research and clinical application is evident in these areas, as scientific findings are rapidly translated into improved patient care.
Feature Traditional Disease Management Precision Medicine Approach One-size-fits-all; population-level guidelines Individualized; tailored to patient’s unique profile Diagnostics Symptom-based; broad lab tests Genomic sequencing; specific biomarkers Treatment Standard therapies; trial-and-error Targeted therapies; personalized drug selection Prevention General lifestyle recommendations Risk stratification based on genetics/lifestyle Focus Treating disease after onset Proactive prevention & early intervention Innovations in Oncology and Urology
Oncology and urology are two fields experiencing rapid innovation, driven by a deeper understanding of disease mechanisms and the development of highly targeted therapies.
In prostate cancer, a common malignancy affecting men, research is continuously refining diagnostic and treatment strategies. Beyond traditional approaches, new insights into genetic predispositions and molecular pathways are leading to more personalized interventions. For example, while issues like Causes of frequent peeing might seem minor, they can sometimes be indicative of underlying urological conditions, including prostate issues, highlighting the importance of thorough diagnostic research.
Immunotherapy has revolutionized cancer treatment, harnessing the body’s own immune system to fight malignant cells. One of the most exciting developments is the use of CAR-T cells, where a patient’s T-cells are genetically engineered to recognize and attack cancer cells. Early clinical trials are showing remarkable success in various cancers, including certain hematological malignancies.
For urologic cancer treatments, research is exploring novel surgical techniques, radiation therapies like proton therapy, and systemic treatments that are less invasive and more effective. The identification of rare genetic syndromes, such as Birt-Hogg-Dubé syndrome (BHD), is also crucial. While phenotypical prevalence is low (1 in 200,000-500,000), studies show that BHD-causing FLCN variants are 75 to 180 times more prevalent in multi-ethnic genomic registry populations. This highlights the importance of genetic screening and early detection screening for at-risk individuals to facilitate timely intervention and management of associated cancers.
These advancements underscore the critical role of ongoing research in transforming outcomes for patients facing urological cancers and other complex conditions.
Cardiovascular and Neurological Progress
The relentless pursuit of knowledge in medical research has led to profound advancements in our understanding and treatment of cardiovascular and neurological diseases, conditions that collectively impact millions globally.
Cardiovascular health remains a cornerstone of public health, with research constantly seeking to mitigate the burden of heart disease and stroke. Atrial fibrillation (AF), affecting over 50 million people worldwide, is a major focus. Recent studies are exploring the use of AI with handheld single-lead ECGs to predict incident AF, offering a potential breakthrough in early detection and prevention. Furthermore, research into the global burden of lead-attributable CVD reveals a significant,, environmental risk factor, prompting calls for public health interventions. Novel therapies like mavacamten are showing promise in treating obstructive hypertrophic cardiomyopathy, a genetic heart condition, by directly addressing the underlying molecular dysfunction.
In neurological research, the focus is on unraveling the complexities of the brain to combat debilitating conditions. The ability to predict dementia up to 16 years before onset using blood biomarkers, as shown in studies from Sweden, represents a significant step forward. This early identification opens a crucial window for potential preventative strategies and clinical trials for diseases like Alzheimer’s. Research into neurodegenerative disorders continues to explore the cellular and molecular mechanisms driving conditions such as Parkinson’s disease and ALS, with the aim of developing disease-modifying therapies.
Improvements in acute care are also critical. For instance, enhanced sepsis management protocols are continually being refined to improve outcomes for both adults and children, leveraging research into optimal diagnostic criteria and treatment pathways. These advancements across cardiovascular and neurological domains exemplify how medical research directly translates into improved longevity and quality of life for patients.
Ethics, Integrity, and Global Health Challenges
The pursuit of medical knowledge, while inherently beneficial, must always be guided by stringent ethical principles and robust regulatory frameworks. This ensures that research is conducted responsibly, safeguarding the rights and well-being of participants and maintaining public trust.
A foundational document in human research ethics is the Declaration of Helsinki, which provides ethical principles for medical research involving human subjects, including research on identifiable human material and data. It emphasizes respect for the individual, their right to self-determination, and the right to make informed decisions to participate in research.
Overseeing these ethical considerations are Institutional Review Boards (IRB), also known as Research Ethics Committees. These independent committees, composed of scientists, ethicists, and community members, play a critical role in reviewing and approving research protocols involving human subjects. Their primary mandate is to protect the rights, welfare, and safety of research participants. They ensure that risks are minimized, benefits are maximized, informed consent is properly obtained, and participant privacy and confidentiality are maintained. This rigorous oversight is a cornerstone of patient safety in medical research.
Despite these safeguards, the field faces ongoing challenges related to research ethics and integrity. The replication crisis, where findings from published studies cannot be reproduced by independent researchers, highlights issues with methodological rigor and reporting. This has led to a greater emphasis on reproducibility sciences and transparent reporting practices to ensure the reliability of scientific discoveries.
Conflicts of Interest and Research Integrity

The financial landscape of medical research, particularly the significant role of industry funding, introduces complex considerations regarding conflicts of interest and their potential impact on research integrity. While industry-funded studies are essential for translating basic science into marketable therapies, the financial ties can create perceived or actual biases.
Industry-funded studies have become a dominant force, funding 62% of biomedical research recently. This substantial investment can sometimes lead to situations where financial interests might unduly influence research design, data interpretation, or publication decisions. For example, studies have shown that industry sponsorship can be associated with more favorable outcomes for the sponsor’s products.
To mitigate these risks, robust mechanisms for transparency and disclosure are critical. Researchers and institutions are increasingly required to disclose all financial relationships with industry, and journals often mandate comprehensive conflict of interest statements. However, the effectiveness of these measures in fully addressing financial bias in biomedical study outcomes remains a subject of ongoing debate.
The integrity of the scientific process relies heavily on rigorous peer review rigor and independent oversight. When conflicts of interest are not adequately managed or disclosed, it can erode public trust in scientific findings and potentially compromise patient care. Striking a balance between leveraging industry resources for innovation and safeguarding the impartiality of research is a continuous challenge for the scientific community.
Addressing Global Resource Constraints
Medical research, while making incredible strides, is confronted by significant global health challenges, many of which are exacerbated by resource constraints in low-income settings. These challenges demand innovative solutions and equitable distribution of research benefits.
One of the most pressing issues is antibiotic resistance. The overuse and misuse of antibiotics globally have led to the emergence of “superbugs” that are resistant to most available drugs. Research into new antibiotics and alternative therapies is critical, but the economic incentives for developing these drugs are often insufficient, particularly for populations in low-income countries where the burden of resistant infections is highest.
The obesity epidemics represent another major global health crisis, contributing to a rise in non-communicable diseases like diabetes and cardiovascular conditions. Research into effective prevention and treatment strategies is vital, but implementing these solutions requires addressing complex socioeconomic and environmental factors, especially in regions with limited public health infrastructure.
In many low-income settings, basic healthcare needs, such as adequate maternal nutrition and access to essential medicines, remain unmet. This directly impacts health outcomes and the capacity for robust medical research. Research efforts must be tailored to these contexts, focusing on cost-effective interventions and building local research capacity.
Furthermore, cancer screening prioritization in resource-limited environments requires careful consideration. Not all screening programs are feasible or beneficial in every setting. Research is needed to identify the most impactful and sustainable screening strategies that align with available resources and local disease burdens. Addressing these global health challenges requires a concerted effort to promote public health equity, ensuring that the benefits of medical research reach all populations, regardless of their economic status.
Conclusion
The landscape of healthcare and medical research is a testament to human ingenuity and our collective commitment to improving quality of life. From the foundational discoveries in basic science to the rigorous validation in clinical trials, every step is a deliberate effort to unravel the mysteries of disease and forge pathways to better health. We have explored the intricate structural framework that guides this journey, the diverse funding mechanisms that fuel it, and the myriad opportunities it presents for the next generation of scientific leaders.
Technological advancements, particularly in AI, CRISPR gene editing, and genomics, are not merely incremental improvements; they are transformative forces, enabling precision medicine and pushing the boundaries of what is curable and preventable. These tools empower us to personalize treatments, predict disease with greater accuracy, and develop novel therapies that target the root causes of illness.
Yet, alongside these triumphs, we acknowledge the persistent challenges: ethical dilemmas in an increasingly complex research environment, the critical need for research integrity amidst financial influences, and the stark realities of global resource constraints. Addressing these issues requires ongoing vigilance, collaborative effort, and a steadfast commitment to equitable access to healthcare advancements.
The impact of medical research on public health outcomes is undeniable. It has led to increased longevity, eradicated diseases through vaccine development, and provided new treatments that have revolutionized the management of chronic conditions. The growing emphasis on Patient and Public Involvement (PPI) further enhances the relevance and quality of research, ensuring that patient voices and lived experiences shape the research agenda.
As we look to the future, the spirit of inquiry, collaboration, and ethical responsibility will continue to drive medical progress for all. The ongoing dedication of researchers, institutions, and policymakers ensures that the promise of better health remains within reach for every individual, everywhere.
Frequently Asked Questions about Healthcare and Medical Research
What are the primary phases of medical research?
Medical research typically progresses through several key phases: basic research (fundamental scientific inquiry), pre-clinical studies (testing in laboratory and animal models), and clinical research (human trials). Clinical research is further divided into phases (Phase 0 to Phase IV), each with specific objectives related to safety, dosage, efficacy, and long-term monitoring.
How does AI improve the accuracy of medical diagnostics?
AI improves medical diagnostics by analyzing vast amounts of data, such as medical images (X-rays, MRIs), pathology slides, and patient records, to identify subtle patterns and anomalies that might be difficult for humans to detect. This leads to earlier and more accurate diagnoses, especially in fields like radiology, pathology, and ophthalmology, ultimately enabling more timely and effective treatment.
What role do institutional review boards play in human trials?
Institutional Review Boards (IRBs) are independent committees that critically review and approve all research protocols involving human subjects. Their primary role is to protect the rights, welfare, and safety of research participants. They ensure that studies are ethically sound, risks are minimized, benefits are justified, informed consent is properly obtained, and participant privacy and confidentiality are maintained throughout the research process.